When people ask whether aluminum rusts, they are usually trying to understand what kind of surface damage aluminum can actually develop in real service conditions. That question matters in construction, transportation, marine equipment, and industrial manufacturing, where corrosion resistance affects both product life and maint
This article explains the difference between rust and aluminum corrosion, what happens when aluminum is exposed to different environments, which corrosion risks matter most, and how aluminum can be protected through the right alloy, finish, and design approach. By the end, readers will have a clearer basis for evaluating aluminum in practical applications.
Quick Answer: Does Aluminum Rust?

No, aluminum does not rust. Rust is a form of iron oxide, and aluminum contains no iron.
What aluminum does form is a thin oxide layer on its surface when exposed to air. In many environments, this layer is stable and protective.
However, aluminum can still corrode under certain conditions, especially when it is exposed to salt, trapped moisture, chemical attack, or galvanic contact.
Why Aluminum Does Not Rust
The key reason aluminum does not rust is simple: it contains no iron.
When fresh aluminum is exposed to oxygen, a microscopic oxide film forms almost immediately. This film acts as a barrier that slows further attack. In many service conditions, that is enough to give aluminum very good corrosion resistance without additional treatment.
Still, this natural protection has limits. In chloride-rich, chemically aggressive, or poorly designed service environments, aluminum can still suffer corrosion over time.
Can Aluminum Corrode?
Yes. Aluminum does not rust, but it can corrode.
The most important point is that aluminum’s natural oxide layer is not invincible. In some service environments, that protective layer can be damaged, penetrated, or destabilized.
The most common risk factors include:
- Chloride exposure, such as seawater or road salt
- Contact with dissimilar metals in the presence of moisture
- Strong acidic or alkaline chemicals
- Trapped moisture in poorly designed joints or crevices
- Inappropriate alloy selection for the service environment
5 Common Types of Aluminum Corrosion
1. Atmospheric Corrosion

Atmospheric corrosion is one of the most common forms of aluminum corrosion. It happens when aluminum is exposed to air, moisture, and pollutants over time.
In normal outdoor conditions, aluminum usually performs well because its oxide layer helps protect the surface. However, in humid, coastal, or polluted environments, long-term exposure can cause dulling, staining, discoloration, or gradual surface attack.
2. Pitting Corrosion
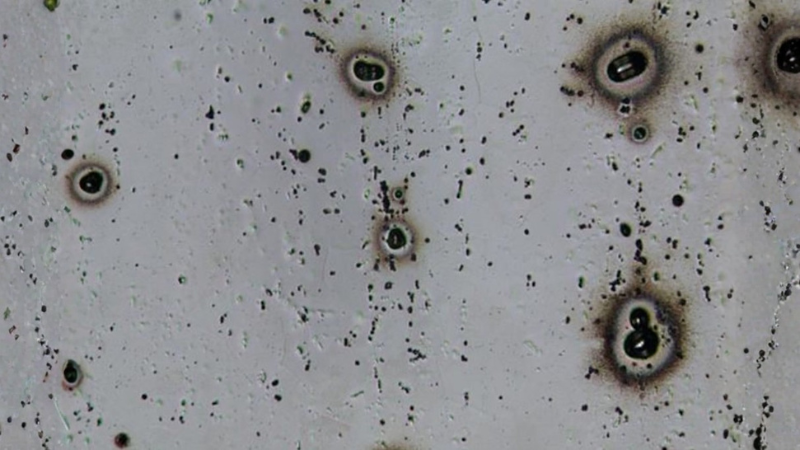
Pitting corrosion is one of the most common forms of aluminum corrosion, especially in chloride-rich environments such as coastal air, seawater exposure, or de-icing salt conditions.
It usually appears as small, localized pits rather than broad, uniform damage. This is why pitting can be difficult to detect early but still become serious in marine, outdoor, or wet service applications.
3. Galvanic Corrosion

Galvanic corrosion happens when aluminum is in electrical contact with a more noble metal, such as stainless steel or copper, in the presence of an electrolyte like water or salt solution.
In that situation, aluminum can become the anodic material and corrode more quickly. This risk is common in mixed-metal assemblies if the joint design, insulation, or fastener choice is poor.
4. Crevice Corrosion

Crevice corrosion may occur where moisture becomes trapped in joints, under gaskets, around fasteners, or in narrow unventilated gaps.
Even when the bulk environment seems mild, stagnant moisture and contamination inside a crevice can create localized corrosion conditions.
5. Chemical Attack
Aluminum can also corrode in aggressive chemical environments. Strong acids and especially strong alkalis can break down the protective oxide layer.
That is why aluminum must be selected carefully for chemical processing, cleaning systems, and industrial environments where the part may be exposed to caustic or acidic media.
How to Protect Aluminum from Corrosion
Aluminum already has a natural defense mechanism, but the right finishing and design choices can improve long-term durability significantly.
Anodizing

Anodizing thickens the natural oxide layer on the aluminum surface through an electrochemical process.
It is commonly used when the part needs:
- better corrosion resistance
- improved wear resistance
- a more controlled decorative finish
- long-term surface stability in architectural or industrial use
Anodizing is widely used on architectural components, consumer products, and precision aluminum parts.
Powder Coating

Powder coating adds a durable barrier layer to the aluminum surface.
It is often chosen when the part needs:
- additional weather resistance
- color and appearance control
- better resistance to surface wear
- a cost-effective protective finish for outdoor applications
It is commonly used on aluminum profiles, outdoor furniture, equipment housings, and automotive or industrial components.
Painting
Painting is another practical barrier method, especially for large fabricated assemblies or parts that may need future repair or touch-up.
Compared with anodizing, painting is often more flexible for maintenance, though long-term performance depends heavily on coating selection, surface preparation, and service conditions.
Proper Alloy Selection

Not all aluminum alloys have the same corrosion behavior. Alloy selection should match the service environment.
For example:
- 5xxx series alloys are often chosen for marine-related applications because of their good corrosion resistance
- 6xxx series alloys are widely used where a balance of strength, formability, and corrosion resistance is needed
If the service environment is demanding, alloy selection should be treated as an engineering decision, not just a purchasing decision.
Good Design and Assembly Practice
Even the right alloy can fail early if the design is poor.
Good corrosion control also depends on:
- avoiding water traps
- reducing tight unsealed crevices
- isolating dissimilar metals where needed
- choosing compatible fasteners
- specifying the right finish for the environment
Aluminum vs Carbon Steel: Corrosion Behavior at a Glance
This does not mean aluminum is always better than steel. It means aluminum is often preferred when corrosion resistance and low weight matter, while steel is often preferred when maximum structural strength or lower raw material cost matters.
Common Applications Where Aluminum’s Corrosion Resistance Matters

Construction
Aluminum is widely used in window frames, curtain walls, roofing components, siding, and architectural trim because it combines corrosion resistance with low weight and good appearance.
Transportation
Aircraft, rail components, body panels, and lightweight vehicle structures often use aluminum because corrosion resistance and weight reduction both matter.
Marine Equipment
Marine-grade aluminum alloys are used for boats, docks, gangways, and other coastal equipment where salt exposure makes corrosion resistance essential.
Consumer and Industrial Products
Electronics housings, outdoor furniture, equipment covers, and appliance components often benefit from aluminum’s ability to resist environmental exposure better than unprotected carbon steel.
Conclusion
So, does aluminum rust? No. Aluminum does not rust because rust is a form of iron oxide, and aluminum contains no iron.
What aluminum does do is form a thin oxide layer that helps protect the surface. That natural protection is one of the reasons aluminum performs well in so many outdoor, architectural, transportation, and industrial applications. But aluminum is not immune to corrosion. Salt exposure, galvanic contact, trapped moisture, poor alloy selection, and aggressive chemicals can still create service problems.
For most buyers and engineers, the real question is not whether aluminum rusts, but whether the alloy, finish, and design are suitable for the environment the part will actually face.
Need Corrosion-Resistant Aluminum Parts? Contact Honjenny
If you need corrosion-resistant aluminum parts for marine, construction, transportation, or industrial applications, send Honjenny your drawing, alloy requirement, service environment, and surface finish standard. Our engineering team can help recommend the most practical material and finishing solution for durability, manufacturability, and cost. Contact Honjenny today to start your inquiry.



